
A body at rest persists in its state of rest, and a body in motion remains in constant motion along a straight line unless acted upon by an external force. The Newton’s three laws of motion are Law of Inertia, Law of Mass and Acceleration, and the Third Law of Motion. In the second law, the force on an object is equal to its mass times its acceleration. In the first law, an object will not change its motion unless a force acts on it. What are Newton’s laws of motion? Newton’s laws of motion relate an object’s motion to the forces acting on it. What is the First and second law of Motion? Second law of thermodynamics is very important because it talks about entropy and as we have discussed, ‘entropy dictates whether or not a process or a reaction is going to be spontaneous’. Why is second law of thermodynamics important? Isolated systems spontaneously evolve towards physics equilibrium, the state with most entropy.
#SECOND LAW OF THERMODYNAMICS IN PHYSICS FULL#
The second law of thermodynamics states that the full entropy of the associated isolated system will ne’er decrease over time, and is constant if and given that all processes are reversible. What is Second Law of Thermodynamics class 11 physics? – Related Questions Which best describes the second law of thermodynamics? The Second Law also states that there is a natural tendency of any isolated system to degenerate into a more disordered state. It states that as energy is transferred or transformed, more and more of it is wasted. What does the second law of thermodynamics state? The Second Law of Thermodynamics is about the quality of energy. The acceleration of an object depends directly upon the net force acting upon the object, and inversely upon the mass of the object. What is the meaning of 2nd law? The second law states that the acceleration of an object is dependent upon two variables – the net force acting upon the object and the mass of the object. What is 2nd law of thermodynamics in physics? The second law of thermodynamics states that heat transfer occurs spontaneously only from higher to lower temperature bodies. The second law also states that the changes in the entropy in the universe can never be negative.
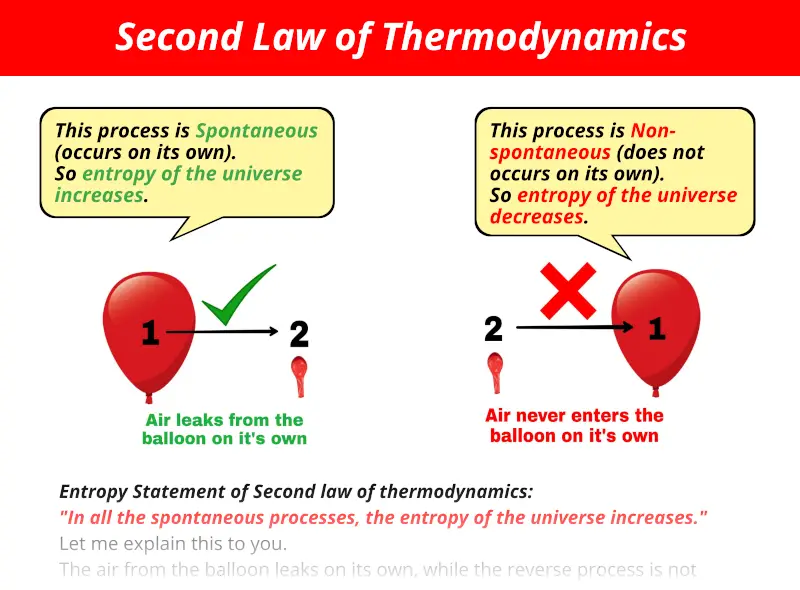
In order for such a process to take place, external work is required.What is Second Law of Thermodynamics class 11 physics? The Second Law of Thermodynamics states that the state of entropy of the entire universe, as an isolated system, will always increase over time. Second law states that such a process is however not spontaneous and natural. It may be noted that, the transfer of heat from a colder body to a hotter body is possible (as in a refrigerator or an air conditioner). It may be proved that all statements of the second law are completely equivalent and revolves around the same central concept of entropy increase. That's what the Clausius' statement is all about. Heat is transferred from a hotter body to a colder body because it is accompanied by entropy increase but, the converse never occurs because as such the entropy of the system is required to decrease. But, no one has ever observed spontaneous transfer of heat from a colder body to a hotter body.Įven though such phenomenon are permissible by the first law, such processes never occur naturally.

Heat always flows from a hotter body to a colder one spontaneously. Natural systems always have a tendency to maximise their entropy.Īnd that's what the second law is all about.Ĭonsider for example, the transfer of heat from one body to another in contact due to the temperature difference. In other words, second law specifies the direction of evolution of a natural process. All irreversible (natural and spontaneous) processes are characterized by the fact that entropy always increases in such processes.Īnd the second law of thermodynamics logically means that entropy always tends to increase.Ī physical system shall always proceed to a state of maximum entropy.
